
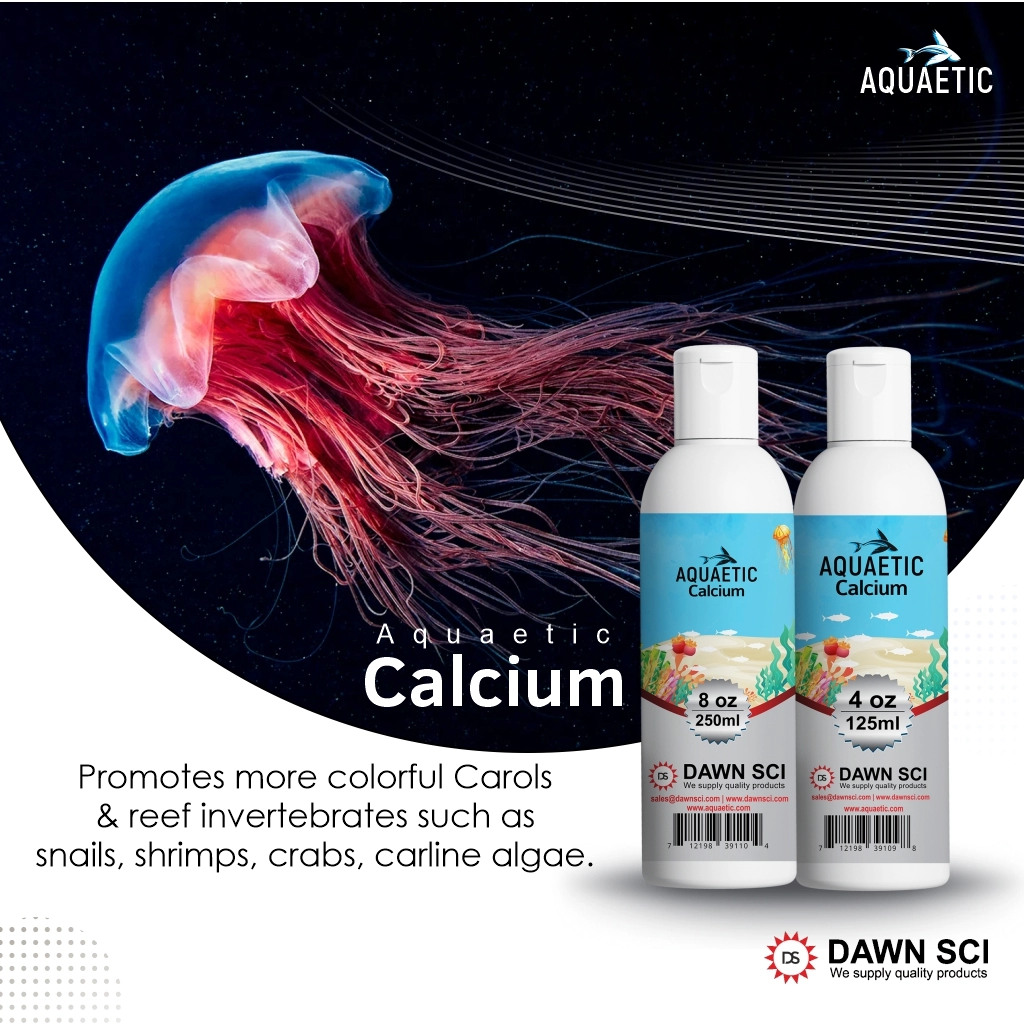
In natural reefs, Calcium Level is very essential, and it is gradually dropping in reef Aquarium as invertebrates utilize the Calcium from water. The Calcium concentration should be 400-500 ppm (mg/L). Aquaetic Calcium is a natural way to increase Calcium levels in your reef Aquarium and should be used whenever Calcium deficiency is present in your water. Use Aquaetic Calcium when water tests show a Calcium level below 400ppm.
- Promotes more colorful Carols and reef invertebrates such as snails, shrimps, crabs, carline algae.
Directions : Add 5 ml. per 10 gallons of Aquarium water whenever you find Calcium level is down to recommended level.
Ingredient : Deionized Water, Calcium Chloride (Calcium Content 3.63-4.30 %)
1 oz can treat up to 60 gallons.
Made in the USA.
Available Pack size
Disclaimer : Any of above statements have not been evaluated by the food and drug administration (FDA).This product is not intended to diagnose, treat, cure, or prevent disease. This information provided is general and should not be taken as medical advice. This product is for research use only.We strongly advise you to have a test batch before use. If you suffer any adverse reactions or symptoms, please discontinue use immediately.








































Reviews
There are no reviews yet.